
Class I medical devices play a crucial role in ensuring patient safety. These devices are often the most basic category of medical equipment, yet their impact is significant. Dr. Sarah Thompson, a leading expert in the medical device industry, states, "Even simple devices must meet strict standards to protect patients." This highlights the essential nature of compliance in delivering safe healthcare solutions.
Many Class I medical devices, such as bandages and sterile gloves, are ubiquitous in healthcare settings. Their reliable use helps prevent infections and supports patient recovery. However, not every manufacturer adheres to the same rigorous safety standards. This variability can lead to questionable quality in some devices.
As the industry evolves, it's vital to reassess the regulations surrounding Class I medical devices. This reevaluation ensures ongoing patient safety and reflects advancements in technology. There's room for improvement in monitoring the performance of these devices. Ultimately, a commitment to quality can enhance the safety of patient care.

Class I medical devices play a crucial role in ensuring patient safety. These devices, which include items like bandages and tongue depressors, may seem simplistic, but they form the backbone of healthcare. According to the U.S. Food and Drug Administration, about 47% of medical devices fall under Class I. Their widespread presence signifies a fundamental layer of care.
Safety is paramount in patient treatment. Class I devices are designed to be low-risk, yet they still require rigorous standards. For instance, the FDA mandates that all Class I devices meet specific performance and safety criteria. This ensures that even everyday items do not compromise patient well-being. An estimated 25 million patients rely on these devices annually, making their effectiveness vital.
However, the industry does face challenges, such as outdated regulations. Some Class I devices may not have changed in years. This stagnation raises concerns about their safety in modern contexts. Continuous review and updates to standards are necessary. Patients deserve the best care, even from basic tools. Maintaining a high standard for Class I devices is key to preventing potential risks and ensuring patient safety every day.
| Device Type | Purpose | Examples | Impact on Patient Safety |
|---|---|---|---|
| Bandages | Wound care and protection | Adhesive bandages, sterile gauze | Prevent infection and facilitate healing |
| Gloves | Infection control | Nitrile gloves, latex gloves | Barrier against contamination |
| Surgical instruments | Perform medical procedures | Scissors, forceps | Enable safe and effective surgeries |
| Dental Devices | Dental treatment and care | Dental trays, orthodontic appliances | Ensure proper dental hygiene and treatment |
| Stethoscopes | Patient monitoring | Acoustic stethoscopes, electronic stethoscopes | Facilitate accurate diagnosis of conditions |
Class I medical devices play a crucial role in patient safety. These devices include items like bandages, examination gloves, and thermometers. They are generally considered low-risk but are essential in everyday healthcare settings. Their functions vary widely, from assisting in basic medical procedures to providing necessary support for patients' needs.
The oversight on Class I devices is important. Manufacturers must ensure these products are safe and effective. While they might not undergo extensive pre-market approval, their compliance with regulations is vital. This can lead to inconsistencies in quality, making some devices less reliable than others. Uncertainties in production protocols might create gaps in safety standards.
Proper training and knowledge about these devices is crucial for healthcare professionals. Understanding how to use them correctly can significantly impact patient outcomes. Issues may arise when staff are not aware of the device's limitations. Regular reviews and updated training could enhance the reliability of these essential tools. Addressing lapses in knowledge and performance can help improve overall patient care.

Class I medical devices play a critical role in ensuring patient safety. These devices include basic items like bandages, tongue depressors, and examination gloves. Though they seem simple, they have to meet strict regulatory standards. Health authorities, such as the FDA in the United States, oversee these devices to ensure they are safe and effective.
The regulatory standards governing Class I medical devices require manufacturers to adhere to quality management systems. This involves documenting and maintaining precise records of materials, manufacturing processes, and testing. Regular audits and inspections are conducted to ensure compliance. However, not all manufacturers consistently meet these expectations. Some may lack the resources or knowledge to implement effective quality controls.
In addition to following guidelines, manufacturers often face financial pressures that can impact product safety. Balancing cost and quality is challenging. Moreover, some devices may receive clearance despite minimal evidence of their effectiveness. This raises questions about how well these regulatory standards protect patient safety. Consumers and healthcare providers must remain vigilant, advocating for continuous improvements in oversight and transparency.
Class I medical devices play a crucial role in ensuring patient safety. These devices include simple items like tongue depressors and bandages. According to the FDA, about 47% of all medical devices fall into the Class I category. They are subject to the least regulatory control, yet they are vital for basic healthcare and preventive measures.
The impact of Class I devices on patient outcomes is significant. A report from the World Health Organization indicates that effective use of basic medical devices can prevent serious complications in 30% of routine procedures. For example, proper wound care can significantly reduce the risk of infection. However, reliance on these devices also presents challenges. Some devices may lack robust quality control, leading to potential safety issues. This raises concerns about their effectiveness, particularly in resource-limited settings where access to more advanced technology is restricted.
Evidently, Class I devices are essential, but they are not without flaws. Studies have shown that improper usage or faulty devices contribute to adverse patient outcomes. The need for ongoing education and training in proper device use cannot be overstated. Improving safety metrics for these devices requires continuous monitoring and feedback from healthcare professionals. This will help optimize patient care and minimize risks associated with basic medical equipment.
Class I medical devices play a crucial role in patient safety by ensuring minimal risks during their use. These devices, which include simple instruments like bandages and tongue depressors, are generally considered to have a lower risk compared to more complex devices. However, they still present potential risks that warrant thorough assessment. For instance, improper use of a device like a thermometer can lead to inaccurate readings, affecting a patient's diagnosis.
Understanding the risks associated with Class I devices is vital for healthcare providers. Even basic devices can cause injury if not maintained or used correctly. A worn-out bandage might not provide adequate protection. Additionally, education and training on these devices can sometimes be inconsistent. This inconsistency may lead to adverse outcomes that could compromise patient safety. Regular audits and assessments are necessary to identify potential hazards.
Healthcare institutions must adopt a proactive stance. Implementing effective monitoring systems can help in detecting issues early on. Involving patients in their care can also enhance safety. They should understand how to use simple devices properly. By fostering an environment of awareness, healthcare providers can mitigate the risks that even Class I medical devices might present. This ongoing assessment of risks ensures that patient safety remains a top priority.
This chart illustrates the various types of risks associated with Class I medical devices and their impact on patient safety. Understanding these risks helps in the assessment and enhancement of safety protocols.
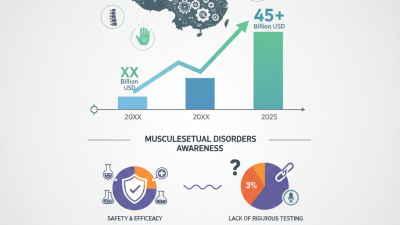




|
This is a medical device. |
