
China's orthopaedic medical devices market is rapidly evolving. It offers a range of products, from implants to advanced surgical tools. Global buyers are increasingly drawn to this dynamic market. They seek high-quality and innovative solutions for various orthopaedic conditions.
One notable aspect of China's orthopaedic medical devices industry is its commitment to research and development. Many manufacturers invest significantly in creating new technologies. This push for innovation helps improve patient outcomes and enhances surgical precision. However, some products still fall short of international standards. This gap raises concerns about quality assurance and credibility.
Furthermore, navigating the regulatory landscape can be challenging for buyers. Understanding the certifications and approvals required for foreign markets is essential. Despite these complexities, the potential for growth in China's orthopaedic medical devices sector is immense. Engaging with local experts can provide valuable insights. This approach helps mitigate risks associated with quality and compliance.

The orthopaedic medical device market in China is expanding rapidly. In 2022, the market size was valued at approximately $7.9 billion, with a projected CAGR of 11.9% until 2027. This growth is driven by an aging population and rising healthcare demands. China's increasing investments in healthcare infrastructure also play a significant role.
Joint replacement devices dominate this market. The demand for orthopedic implants is significant, with hip and knee replacements leading the way. About 1.2 million hip replacements are performed annually in China. However, the market faces challenges. Quality control and regulation are crucial concerns. Many small manufacturers struggle to meet stringent standards, leading to inconsistencies.
Advanced technologies like 3D printing and robotics are emerging trends. These innovations enhance precision in surgeries, improving patient outcomes. They can make procedures less invasive, but awareness and training remain issues. Proper implementation is essential to maximize benefits and mitigate risks. Overall, the landscape is dynamic but requires careful navigation to succeed.
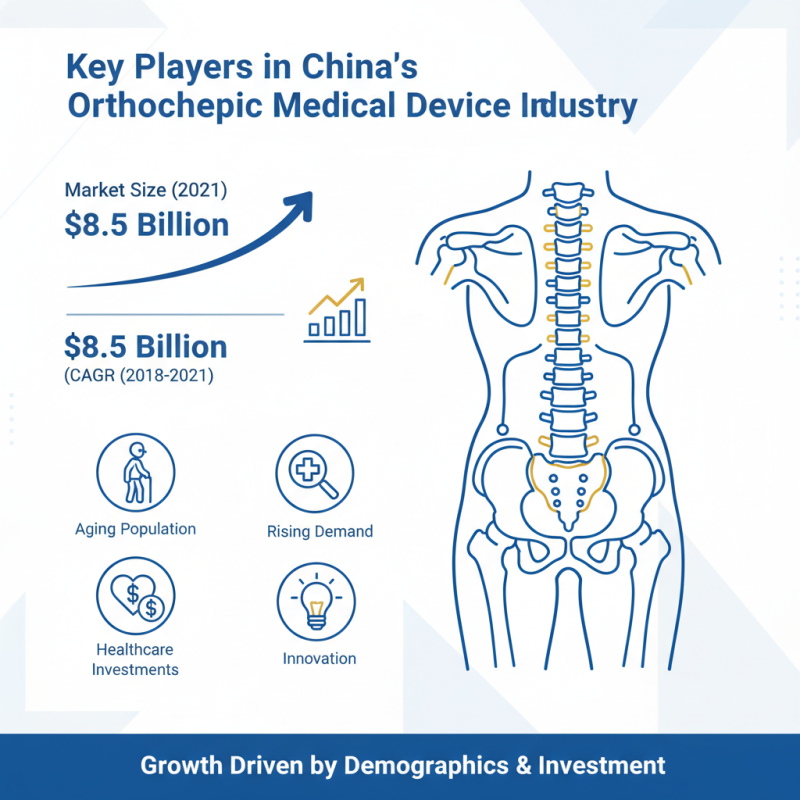
China's orthopaedic medical device industry has seen remarkable growth in recent years. In 2021, the market size reached approximately $8.5 billion, reflecting a compound annual growth rate (CAGR) of about 12%. This growth can be attributed to an increasing elderly population and rising demand for effective treatment options. Healthcare investments have further stimulated innovation within the sector.
Several key players dominate this burgeoning market. Local manufacturers are gaining traction, bolstered by government initiatives that encourage domestic production. Emerging companies innovate rapidly, focusing on products such as implants and surgical instruments. Statistics indicate that around 70% of the market is occupied by domestic firms, hinting at a shift in reliance from international to local products.
However, challenges remain. Some manufacturers struggle with quality control and regulatory compliance. The fast-paced environment can lead to oversights, potentially affecting patient outcomes. Addressing these issues is crucial for sustaining growth and enhancing the credibility of the orthopaedic medical device industry in China.
China is emerging as a leader in the orthopaedic medical devices sector, driven by innovative technologies. Advanced materials, such as bio-compatible polymers, enhance implant durability. Moreover, 3D printing is revolutionizing custom implants, catering to individual patient anatomy. This personalization improves surgical outcomes and patient satisfaction.
Robotics also play a pivotal role in orthopaedic surgeries. Surgical robots provide precise movements, reducing recovery times. However, there are challenges. The cost of implementing these technologies can be high. Additionally, training surgeons to use advanced equipment is essential. Implementing these solutions requires a fine balance between innovation and practical application.
Telemedicine has gained traction in orthopaedic care. Remote consultations and follow-ups allow patients to receive timely advice. But connectivity issues in rural areas can hinder access. It's a reminder that while technology offers solutions, equitable access remains a critical issue. The landscape is evolving, but there are still important hurdles to overcome.
China's regulatory environment for orthopaedic devices plays a crucial role in the global market. The National Medical Products Administration (NMPA) oversees the approval processes. This body enforces strict guidelines to ensure device safety and effectiveness. Manufacturers must navigate a complex landscape of regulations. Changes can occur rapidly, creating uncertainty.
The approval pathway can involve lengthy documentation and evaluation processes. New entrants face challenges in understanding local requirements. The need for clinical data often complicates matters. As a result, obtaining regulatory approval can be a daunting task for foreign companies. Adapting to evolving standards requires constant vigilance and adjustments in strategy.
Despite these hurdles, opportunities abound in China's orthopaedic sector. The growing elderly population increases the demand for joint replacements and surgeries. Companies must stay informed about regulatory updates to enhance their competitiveness. Collaborating with local partners can ease the navigation of this intricate system. Balancing compliance with market needs is essential for success in this dynamic field.
| Device Type | Usage | Regulatory Status | Market Demand | Growth Forecast |
|---|---|---|---|---|
| Orthopaedic Implants | Bone fixation and repair | Approved by NMPA | High demand in urban hospitals | Expected 8% annual growth |
| Surgical Instruments | For various orthopaedic surgeries | Approved by NMPA | Growing demand in surgery centers | 6% annual growth forecast |
| Orthotic Devices | Support and correct musculoskeletal issues | Registered with NMPA | Increasing use in rehabilitation | 5% growth rate anticipated |
| Joint Replacement Components | Knee and hip replacements | Approved by NMPA | High demand with aging population | 10% annual growth forecast |
| Diagnostic Imaging Devices | MRI and CT scans for musculoskeletal evaluation | Certified under NMPA regulations | Consistent growth in healthcare facilities | 7% growth expected |

The global demand for orthopaedic medical devices continues to rise, creating significant export opportunities for Chinese manufacturers. According to a recent report by the Global Industry Analysts, the orthopaedic devices market is projected to reach USD 60 billion by 2027. China, as one of the fastest-growing producers, plays a vital role in this growth. However, challenges persist, including quality control and adherence to international standards.
Chinese orthopaedic device exports topped USD 3 billion in 2022, reflecting a steady increase in global market share. Key markets include the United States, Europe, and Southeast Asia, where unmet needs drive demand. Yet, many products struggle with international regulatory scrutiny. Reports indicate that overcoming these barriers requires continuous innovation and a commitment to quality assurance.
Manufacturers must invest in R&D to enhance product reliability. The rise of advanced technologies, such as 3D printing for customised implants, holds promise for improving competitiveness. Nevertheless, balancing cost reduction with quality enhancement remains a critical challenge. This dynamic landscape presents opportunities and hurdles in positioning Chinese orthopaedic medical devices in the global arena.





|
This is a medical device. |
